Why does fiber reactive dye work the best on cotton?
Why does fiber reactive dye work the best on cotton?
Name: Delia
Country or region: US
Message: WHY does fiber reactive dye work the best on cotton? I know how fiber reactive dye works and how cotton is made and that it is a cellulose material, but WHY does it work best on cotton? That is what I need to find out. THANKS A MILLION!!!!!!!!!!!
When somebody says that fiber reactive dyes work best on cotton, they're comparing cotton to synthetic fibers, or to dyeing a cotton/polyester blend. Polyester does not react with fiber reactive dyes at all, so they just wash out of it. As a result, dyeing a cotton/polyester blend results in much paler, less satisfactory colors than dyeing 100% cotton.
Fiber reactive dyes don't work better on cotton than on certain other fibers. Fiber reactive dyes also work very well on silk. In addition, processed cellulose fibers, in the form of rayon (also known as viscose), dye more brightly than unmercerized cotton, because the cellulose is more readily available to react with the dye. So, you could say that fiber reactive dyes work best on rayon, unless you're concerned about the way rayon wears more quickly in the laundry.
(Mercerized cotton dyes more brilliantly than unmercerized cotton, because the mercerization process removes small imperfections in the surface that scatter light, causing the color of the fiber to appear lighter and less intense. So, mercerized cotton dyes better than regular cotton. You can often buy mercerized cotton clothing for dyeing.)
The reason why fiber reactive dyes such as Procion MX dyes react well with cellulose, under high-pH conditions, as in the presence of sodium carbonate (soda ash), is that the cellulose forms an anion, as the result of depronotation by the high pH. The cellulosate anion can then attack the dye ring at the carbon atom that holds one of the chlorine atoms. The halogen makes the ring susceptible to this attack. As explained by John Shore in the book Cellulosics Dyeing,
Fiber reactive dyes don't work better on cotton than on certain other fibers. Fiber reactive dyes also work very well on silk. In addition, processed cellulose fibers, in the form of rayon (also known as viscose), dye more brightly than unmercerized cotton, because the cellulose is more readily available to react with the dye. So, you could say that fiber reactive dyes work best on rayon, unless you're concerned about the way rayon wears more quickly in the laundry.
(Mercerized cotton dyes more brilliantly than unmercerized cotton, because the mercerization process removes small imperfections in the surface that scatter light, causing the color of the fiber to appear lighter and less intense. So, mercerized cotton dyes better than regular cotton. You can often buy mercerized cotton clothing for dyeing.)
The reason why fiber reactive dyes such as Procion MX dyes react well with cellulose, under high-pH conditions, as in the presence of sodium carbonate (soda ash), is that the cellulose forms an anion, as the result of depronotation by the high pH. The cellulosate anion can then attack the dye ring at the carbon atom that holds one of the chlorine atoms. The halogen makes the ring susceptible to this attack. As explained by John Shore in the book Cellulosics Dyeing,
"[Reactive dyes] based on nitrogen-containing heterocyclic rings bearing halogeno substituents undergo nucleophilic substitution. The heteroatoms in the aryl ring activate the system for nucleophilic attack because of their electronegativity. The attacking neutrophile can be either a cellulosate anion or a hydroxide ion, the former leading to fixation on the fibre and the latter resulting in hydrolysis of the reactive dye."
Here is a link to a picture I adapted from one in Shore's book, showing the reaction between a Procion MX type dye and a cellulose molecule (click on this image to see the full-sized picture):
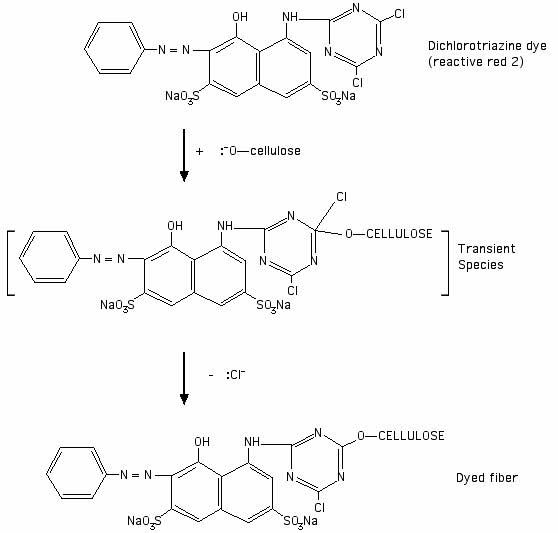
A synthetic fiber such as polyester cannot form an anion that would attack the reactive part of a fiber reactive dye molecule. The type of dye suitable for dyeing polyester, known as Disperse dye, lacks any reactive site for the cellulose in the cotton to attack.
On the other hand, you might be asking an entirely different question. Instead of why do fiber reactive dyes work the best on COTTON, that is, why is this fiber best for these dyes, you might be asking why FIBER REACTIVE dyes work best on cotton, that is, why this type of dye works better than others, for this fiber. Whether or not they work best depends on your priorities. If you want a dye that will not bleed or wash out, even when washed in boiling water, then fiber reactive dyes are the best, because they form a permanent covalent chemical bond to the fiber. However, if your priority is a dye that will not fade in sunlight, you're often going to be better off with vat dyes, instead of fiber reactive dyes, because vat dyes are the class of dyes that is most resistant to damage from light. The most widely available dye, all-purpose dye, such as Rit brand dye, is inferior to both fiber reactive dyes and vat dyes, since it washes out far more quickly, and does not last longer when exposed to bright light.
Further reading:
Posted: Wednesday - October 13, 2010 at 06:50 AM
Follow this blog on twitter here.
Quick Links
- All About Dyes & Dyeing Top -
- Top of this blog -
- FAQ -
- The Dye Forum -
- How to Tie Dye - How to Batik -
- Books - Toys - Plants -
- Top of this blog -
- FAQ -
- The Dye Forum -
- How to Tie Dye - How to Batik -
- Books - Toys - Plants -
More in this category:
- -
Statistics
Total entries in this blog:
Total entries in this category:
Published On: Aug 29, 2012 02:49 PM
Total entries in this category:
Published On: Aug 29, 2012 02:49 PM