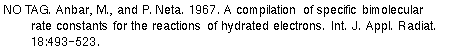
, and is thus a poor OH. scavenger, did not inhibit the DNA strand scission reaction at all.
Nicking of DNA is a good assay for hydroxyl radical production because the hydroxyl radical reacts quite rapidly with DNA,67 causing single-stranded scission,49 but it is unlikely that the attack of singlet oxygen on DNA will give rise to either single stranded breaks or alkali-labile bonds.223 The main value of DNA strand scission as an assay, however, is its high degree of applicability to the situation inside a cell undergoing photodynamic attack.

In order to compare a reaction in water to the same reaction in deuterium oxide, every solution had to be made up from anhydrous reagents in deuterium oxide (Sigma 99.8% D2O). It was necessary to use the formula
actual pH of D2O solution = apparent pH - 0.4
in order to prepare buffers of comparable acidity. DNA was ethanol-precipitated and then redissolved in either aqueous phosphate buffer or phosphate buffer prepared in D2O before its use in this experiment.
The single-stranded nicking of isolated pBR322 DNA was dependent on the presence of three reactants: dye, added iron, and a reductant such as NADH. As all reactions contained EDTA as provided by the DNA as supplied, it was not necessary to add a chelator to see nicking as long as other required
reactants were provided. When the DNA was illuminated in the absence of these components (fig. 46), only a small degree of nicking was seen, present in the DNA mixture as supplied. When 400 mm NADH and 25 mm iron (III) were added, nicking was increased to a small degree, around 13%. In the absence of NADH but presence of iron and 5 mm azure c, nicking was around 10%. Without added iron, NADH plus dye yielded 37% nicking, due probably to contaminating trace metals in the DNA as supplied; with iron, nicking increased to 100%.
This DNA nicking was inhibitable by catalase, SOD, and the OH. scavengers
dimethyl sulfoxide, ethanol, benzoate, and thiourea. Thus the DNA strand scission
was prevented either by scavenging the essential reactants of the iron catalyzed
Haber-Weiss reaction, O2- and H2O2, or by scavenging the ultimate product of the
reaction, OH.. Catalase had a stronger inhibitory effect than did SOD; I believe that
the lower level of inhibition by scavenging of O2- reflects the ability of the excited dye
radical to substitute for O2- in the reduction of chelated ferric iron in the
iron-catalyzed Haber-Weiss reaction. The addition of similar concentrations of
bovine serum albumin did not inhibit the nicking, thus demonstrating that inhibition of
nicking is not a general protein effect. Urea, which reacts with OH. more slowly than
the other scavengers by a factor of 104
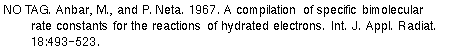
, and is thus a poor OH. scavenger, did not inhibit the DNA strand scission reaction at all.
Similar results were seen for other dyes, including the other thiazines, thionin (fig. 47), toluidine blue o (fig. 48), and methylene blue (fig. 49); the
acridines, acridine yellow (fig. 50), acridine orange (fig. 51), and proflavin (fig. 52); the xanthenes, rose bengal (fig. 53) and fluorescein (fig. 54); the naphthalimide lucifer yellow (fig. 55); and hematoporphorin derivative (fig. 56). All of these dyes followed the same pattern of less damage in the absence of reductant or in the presence of DMSO or thiourea, without significant decrease in damage conferred by urea. The substituted acridine quinacrine did no damage to the DNA (fig. 57). Control experiments in a previous paper200 show that OH. scavengers do not interact directly with O2- or H2O2, and they do not slow the oxidation of NADH or glutathione, or the reduction of cytochrome c by illuminated dyes in the presence of the reductant.
Increasing concentrations of azure c led to increasing levels of DNA damage in the presence of NADH and iron (fig. 58). For NADH levels of 0.4 mM, the damage produced by the azure c leveled off after the concentration of azure c reached 8 mM.
Different quantities of DMSO were added to a reaction including 5 mM azure c and 0.4 mM NADH (fig. 59, panel A). Increasing quantities prevented more and more of the DNA strand scission from occurring, as one would expect, until essentially all of the reaction was blocked at 1 M. When the log of the % nicking is plotted against the log of the DMSO concentration (fig. 59, panel B), a nearly straight line is obtained, showing the competition of the DMSO with the DNA for the agent which causes the DNA nicking, OH..
The level of iron present in the reaction also affected the degree of DNA strand scission seen (fig. 60). Increasing the concentration of the FeCl3 dramatically increased the degree of DNA damage. This produced one difficulty
.4 mM; FeCl
in these experiments. The DNA as supplied contained different amounts of iron, and contained the chelator EDTA; however, except in the experiment comparing the solvent deuterium oxide to water, the DNA was used without dialysis or precipitation, in order to minimize preliminary strand scission of the DNA, so that iron contamination was not controlled for. The experiments shown in figures 66 and 68 were performed for shorter periods of illumination, to counter the increased amounts of damage seen in the reactions, evidently due to contamination with higher concentrations of iron in the Boehringer Mannheim DNA used in these experiments and in the experiment shown in figure 69, as opposed to the Pharmacia DNA used in the other experiments. Furthermore, the experiment shown in figure 65 showed more damage than would have been expected in the reactions performed in the absence of NADH, compared to similar experiments represented in other figures, because the Boehringer Mannheim DNA was used without adjusting the length of incubation accordingly.
Iron is not the only metal that can act as a catalyst for the Haber-Weiss reaction (fig. 61).44 Copper can, as well, although the effects of chelators are different. The lower band in each lane in the photograph contains supercoiled DNA, while the upper band contains nicked DNA; linearized DNA would occupy a band in a position between these two bands, and highly degraded DNA a smear. Lane one shows the DNA largely nicked after treatment with azure c, NADH, EDTA, and iron. Lane two shows a similar treatment, but with cupric chloride substituted for the iron, resulting in a smear of degraded DNA. Lane three shows that additional EDTA did not much affect the iron reaction, as the DNA looks much the same as in lane one. Lane four shows that additional
Figure 61. Nicking of DNA in the presence of different metals and chelators. All reactions contained 7.5 mg/ml DNA, 0.4 mM NADH, 5 mM azure c, and 12.5 mM EDTA in 100 mM phosphate buffer, pH 7.6. From left to right, the reaction whose results are shown in lane 1 contained 12.5 mM added FeCl3 and 12.5 mM EDTA; lane 2, 12.5 mM cupric chloride and 12.5 mM EDTA; lane 3, 12.5 mM added FeCl3 and 37.5 mM EDTA; lane 4, 12.5 mM cupric chloride and 37.5 mM EDTA; lane 5, 12.5 mM added Fe, 12.5 mM EDTA, and 25 mM deferoxamine; lane 6, 12.5 mM added Fe, 12.5 mM EDTA, and 25 mM DTPA; and lane 7, 12.5 mM added Fe, 12.5 mM EDTA, and 25 mM citrate.
EDTA partly inhibited the copper's ability to catalyze the production of OH.. (Lane five shows that the addition of the chelator deferoxamine, a chelator
known for its ability to bind iron in such a way that it cannot catalyze the Haber-Weiss reaction, almost completely prevents DNA damage. Lanes five and six show that DTPA and citrate lack this ability, or are unable to competetively sequester the Fe3+ from the EDTA that is present.)
The requirement for NADH in order to see substantial amounts of DNA strand scission varied from dye to dye (fig. 63). Acridine orange, methylene blue, and proflavin produced the most substantial amounts of strand scission in the absence of substrate; azure c, rose bengal, and neutral red produced far less damage without an oxidizable substrate. In most cases, however, the OH. scavenger DMSO prevented most of this damage, and in all cases it had some inhibitory effect, even in the absence of oxidizable substrate. While the amount
mM; proflavin, 10 mM; azure c, 10 mM; DNA, 7.5 mg/ml; NADH, 2 mM; FeCl
of DNA damage increased substantially in the presence of NADH, an even greater proportion of the total damage was prevented by the OH. scavenger DMSO. The presence of oxidizable substrate is important in deciding whether a type I or type II reaction will predominate. Reactions occurring without substrates but with dyes may have been caused by direct DNA oxidation by triplet state dye or by the oxidation of dye to a more positive valence state with the release of an electron to O2 to form O2- in a type II oxidation (see Introduction).
DNA strand scission could be accomplished with a range of substrates in addition to NADH (fig. 64). (As in figure 61, the lower band in each lane in the photograph contains supercoiled DNA, while the upper band contains nicked DNA.) Increasing concentrations of each of the oxidizable substrates resulted in increasing amounts of DNA strand scission. Glutathione at 0.4 mm was as effective in the reaction as the same concentration of NADH, in the presence of 25 mm added iron(III); 1 mm of either substrate resulted in complete strandscission when exposed to 5 mm azure c. Tyrosine and tryptophan, which were tested in the presence of 50 mm iron, were also capable of acting as substrates, and gave increasing amounts of DNA strand scission as their concentrations were increased. DMSO at a concentration of 1.4 m completely inhibited the single stranded DNA nicking, suggesting once again that OH. are the mediators of damage.
Not all dyes reacted equally with the different substrates. In the presence of sufficient dye to result in 100% strand scission with NADH, azure c showed a marked degree of damage with tryptophan and tyrosine, while rose bengal,
Figure 64. Increasing concentrations of substrates have increasing effects. All reactions contained 5 mm azure c and were illuminated for 75 minutes. NADH and glutathione reactions contained 25 mm added iron (III); tryptophan and tyrosine, 50 mm added iron (III). Each reaction contained 125 ng of supercoiled pBR322 DNA dissolved in 20 ml phosphate buffer with 12.5 mm EDTA.
proflavin, and neutral red gave little, if any, more damage in the presence of tryptophan or tyrosine than in the absence of any oxidizable substrate (fig. 65). When used in place of NADH, GTP increased the amount of DNA damage over that seen without an oxidizable substrate particularly for the dye rose bengal, but also for methylene blue and azure c (fig. 66). For the dyes neutral red, proflavin, and fluorescein, however, GTP had no such effect; it acted only as a non-specific scavenger, instead (its reaction rate with OH. is 7.6 x 109).92 When the damage resulting from the reaction of rose bengal with GTP was studied (fig. 67), the OH. scavenger DMSO was found to lend a protective effect, but the enzymes catalase and superoxide dismutase were not significantly protective, as they were in the presence of NADH, suggesting that this strand scission reaction proceeds via a different mechanism than that characteristic of the other substrates. Hydroxyl radical scavengers were far more protective against damage than were the enzymes SOD and catalase for the reactions between dGMP and rose bengal and between dGMP and azure c, as well (fig. 68).
.4 mM; tyrosine, 4 mM; tryptophan, 4 mM; FeCl
of damage was seen, instead (fig. 69). Inhibition of the reaction suggests that a protonation step, which is slower when the deuterium ion must be exchanged instead of the hydrogen ion. Protonation is an obligate part of two of the reactions that leads to the formation of OH. from superoxide.
O2- + O2-+ 2H+ --> H2O2 + O2
Fe2+EDTA + H2O2 + H+ --> OH. + H2O
Moreover, 1.4 m DMSO protected against strand scission even in the presence of deuterium oxide. Since DMSO reacts very slowly with singlet O2 but very rapidly with OH., it is clear that deuterium is inhibiting free radical reactions leading to OH. production and DNA strand scission and not a reaction involving singlet oxygen as an essential intermediate. Interestingly, a similar experiment performed in the absence of oxidizable substrate gave the same result-inhibition of DNA damage by deuterium oxide.
scission, including the thiazines azure c, methylene blue, thionin, toluidine blue o, the xanthenes rose bengal and fluorescein, the acridines proflavin, acridine yellow, and acridine orange, as well as the phenazine neutral red, the naphthalimide lucifer yellow, and the metabolite derivative known as hematoporphyrin derivative. DNA strand scission appears to be an even more sensitive assay for hydroxyl radical production than either the salicylate assay or the thiobarbiturate assay. As was the case for hydroxyl radical production, DNA strand scission in the presence of dye was increased by the presence of an oxi-
dizable substrate. Appropriate compounds included NADH, glutathione, tryptophan, tyrosine, GTP, and dGMP. Scavengers including catalase, SOD, thiourea, formate, azide, benzoate, ethanol, and DMSO prevented damage to the DNA. The reason for SOD's protective effect being so much greater than was seen in the hydroxyl radical assays is presumably the lower light level used in these experiments; the dyes, when excited by light, are reduced by the substrates and then react with oxygen to produce superoxide in either case; when the light is very bright, perhaps the supply of oxygen is a limiting factor, leaving semireduced dyes to react with the oxidized iron, while in dim light, oxygen reacts with most of the smaller amount of semi-reduced dye that is produced, leaving less to reduce the iron. In a very important experiment in this chapter, the deuterium oxide effect showed that, whether or not singlet oxygen is produced in quantity by the illuminated dyes, it is not responsible for the DNA damage they cause. Moreover, even the non-intercalating xanthenes nicked DNA, showing that intercalation is not essential for DNA damage by dyes. Hydroxyl radical mediates the DNA nicking effect of illuminated dyes.